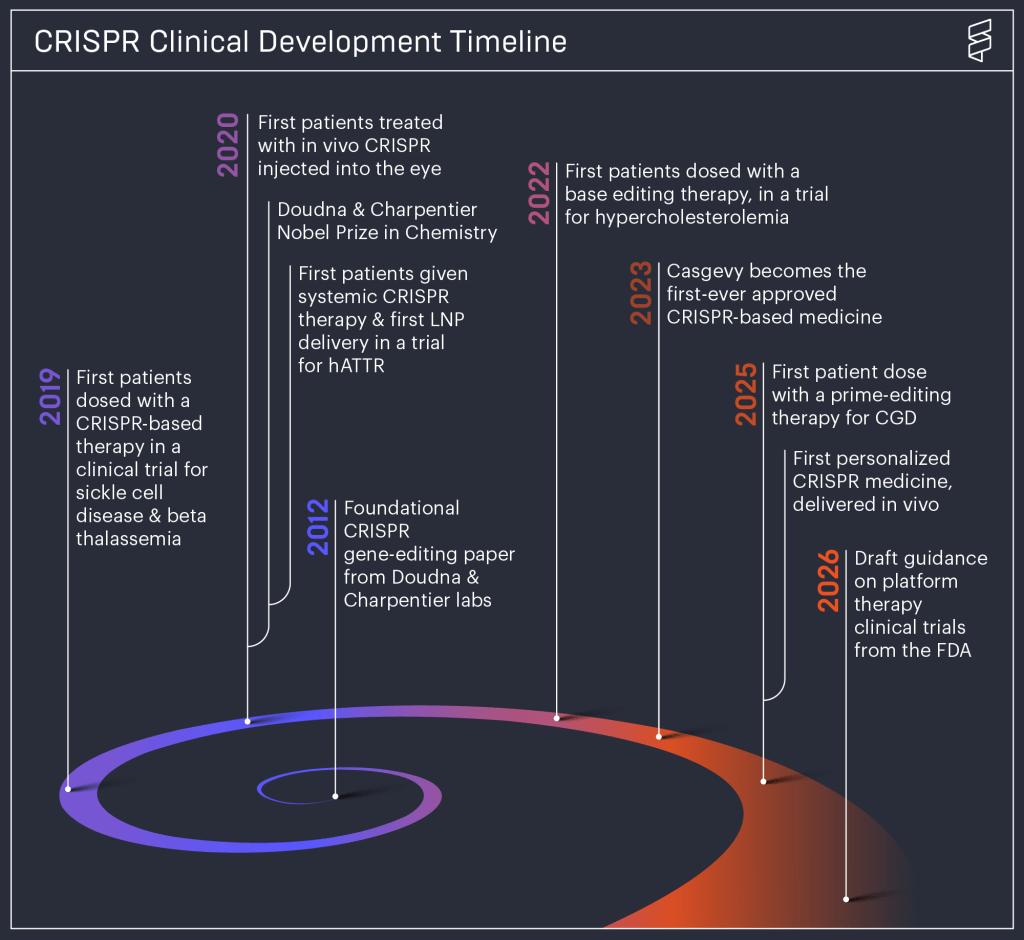
Craig Venter was the first person to read his own genome (his full sequence was published in 2007). He was able to study his genetic predispositions and undergo the most advanced tests to verify their real-world relevance. In 2014, he launched a company called Human Longevity with the goal of building bridges between genetic sequences and diagnosis. In this way, by 2016 he had identified and defeated prostate cancer, but science and luck were not enough to save him a second time in 2026. When he passed away on April 29, he had not accomplished everything he had hoped to, but more than enough to secure a place in history, and perhaps even earn the respect of many of his former rivals.
Continue reading