
Credit: Keegan Houser/UC Berkeley
Interview given to Anna Meldolesi (Corriere della sera, 15 May 2018)
The CRISPR biomedical duel between China and the US has been called “Sputnik 2.0”. Is Europe being left behind?
JD: As with any disruptive technology, there is intense competition to lead. However, unlike the space race, the CRISPR research effort is global and more collaborative. We consistently see key advances in CRISPR technology shared through scientific papers, written and read by research teams around the world. This collective approach has helped to democratize the technology. However, differing regulations across countries may impact how we ultimately translate research into real-world applications that can benefit the most number of people with the most need. Researchers in Europe have made valuable contributions to the development and application of CRISPR and will continue to play a role in establishing global standards. Continue reading →




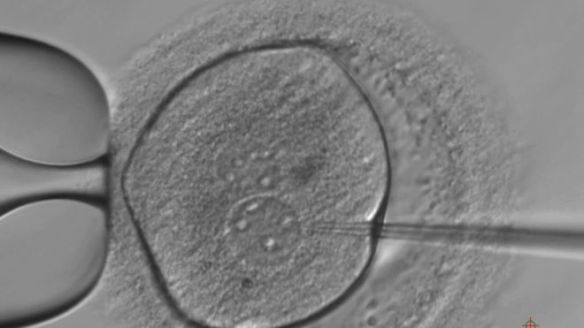 They are the first human embryos edited in Europe and reported in scientific literature. The key difference with experiments already carried out in China and US is that the research published by
They are the first human embryos edited in Europe and reported in scientific literature. The key difference with experiments already carried out in China and US is that the research published by  Biodiversity is a wonderful interplay between genetics and evolution, and butterflies are a fascinating example with their variety of patterns and colors. Understanding how the same gene networks engender visual effects so diverse in thousands of Lepidoptera species is a longtime ambition for many entomologists and evolutionary biologists. The good news is that scientists nowadays have a straightforward technique working with organisms that were difficult to manipulate with conventional biotech tools. Obviously, we are talking about CRISPR. Two papers published in PNAS last week describe how genome editing was used to alter the genetic palette of colors in butterflies and how their wings changed as a result. We’ve asked the entomologist Alessio Vovlas, from the Polyxena association, to comment these stunning experiments.
Biodiversity is a wonderful interplay between genetics and evolution, and butterflies are a fascinating example with their variety of patterns and colors. Understanding how the same gene networks engender visual effects so diverse in thousands of Lepidoptera species is a longtime ambition for many entomologists and evolutionary biologists. The good news is that scientists nowadays have a straightforward technique working with organisms that were difficult to manipulate with conventional biotech tools. Obviously, we are talking about CRISPR. Two papers published in PNAS last week describe how genome editing was used to alter the genetic palette of colors in butterflies and how their wings changed as a result. We’ve asked the entomologist Alessio Vovlas, from the Polyxena association, to comment these stunning experiments.  The University of Berkeley has opened a glimpse into the way bacteria use CRISPR, the microbial immune system that inspired the invention of the method for genetic modification also known as CRISPR. The paper published in
The University of Berkeley has opened a glimpse into the way bacteria use CRISPR, the microbial immune system that inspired the invention of the method for genetic modification also known as CRISPR. The paper published in  Mosquito nets are not enough, vaccines are late to come, land reclamation in Africa is a challenge. But there is a new hope for defeating malaria, coming directly from the most advanced CRISPR frontier. The trick is a kind of genetic chain reaction fuelled by genetic elements called “gene drives”. Researchers are experimenting their power with the aim of crashing the number of mosquitoes responsible for Plasmodium transmission, by spreading genes that are bad for Anopheles gambiae. A gene behaving in Mendelian way has a 50% chance of being passed on from parent to offspring, but it can virtually reach 100% with a little help from a drive. Thus a gene designed to damage a harmful species can propagate within a few generations with a domino effect, until the population collapses. One of the founders of this futuristic strategy is an Italian molecular parasitologist:
Mosquito nets are not enough, vaccines are late to come, land reclamation in Africa is a challenge. But there is a new hope for defeating malaria, coming directly from the most advanced CRISPR frontier. The trick is a kind of genetic chain reaction fuelled by genetic elements called “gene drives”. Researchers are experimenting their power with the aim of crashing the number of mosquitoes responsible for Plasmodium transmission, by spreading genes that are bad for Anopheles gambiae. A gene behaving in Mendelian way has a 50% chance of being passed on from parent to offspring, but it can virtually reach 100% with a little help from a drive. Thus a gene designed to damage a harmful species can propagate within a few generations with a domino effect, until the population collapses. One of the founders of this futuristic strategy is an Italian molecular parasitologist:  The aim is engaging: to treat an increasing number of diseases by correcting the underlying genetic defects. And researchers are breathing optimism at last. The San Raffaele Telethon Institute for Gene Therapy (
The aim is engaging: to treat an increasing number of diseases by correcting the underlying genetic defects. And researchers are breathing optimism at last. The San Raffaele Telethon Institute for Gene Therapy (