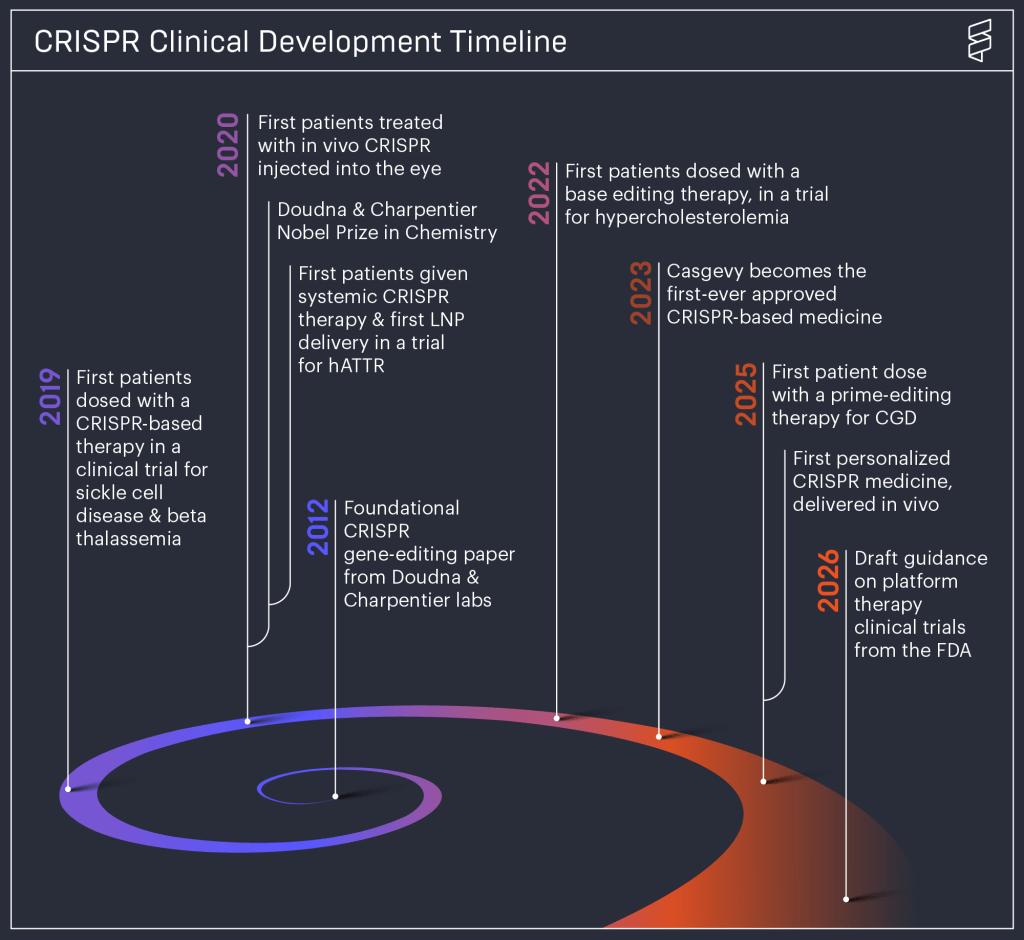
Four years after an experimental treatment for a rare disease, the child underwent surgery for cancer, the first case caused by a virus widely used as a vector, but he is now doing well.
The question of whether gene therapy using adeno-associated viruses (AAVs) can cause tumors has been the subject of intense debate over recent decades. Until now, our understanding of their mechanisms and the data collected over the years from thousands of patients had appeared reassuring. In fact, AAVs are the most commonly used vectors for “in vivo” therapies precisely because they usually deliver their package, containing the therapeutic gene, near the patient’s DNA without integrating into it, thereby avoiding disruptions. Will the risk-benefit balance now change, after the first documented case of a tumor linked to their use in a clinical trial?
Continue reading