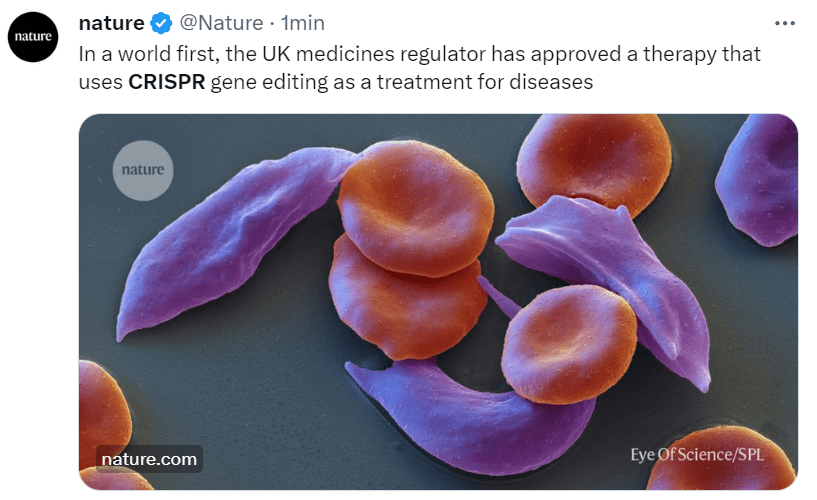
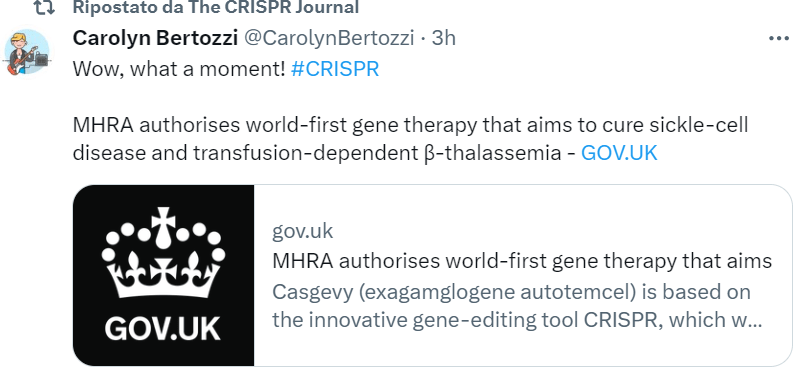
A wait-and-see atmosphere lingers in the biotech and pharma sector. Vice President-designate J.D. Vance recently spoke with enthusiasm on Joe Rogan’s podcast about the first CRISPR treatment to hit the market—the one for sickle cell anemia. However, there’s rising concern about RFK Jr., who could exert significant influence over health and food policies. His broad “natural = good” ideology is both philosophically and scientifically dubious, and his clear anti-GMO positions are worrisome. Listening to his conversation with anti-biotech activist Jeffrey Smith on the RFK Jr. podcast reveals a revival of the classic 1990s narrative—corporations as villains, a corrupt establishment, suppressed scientists, concealed health risks—all now repurposed to criticize CRISPR technology, which is portrayed as unreliable and uncontrollable.