
Italy has a strong tradition in malaria research, and it’s noteworthy that one of the most innovative tools to fight this disease in the future was developed with the crucial contribution of an Italian scientist. Andrea Crisanti has become a familiar face in the country due to his expertise in COVID-19 epidemiology and numerous TV appearances, first during the pandemic and later in his role as a senator. Yet his international reputation is more firmly rooted in a different field: the development of CRISPR-based strategies to eliminate malaria-transmitting mosquitoes.
Continue reading





 Choose a word to fill the gap in the sentence. “Gene drives are an ambitious experiment in …”. Genetics? Ecology? Evolution? Obviously,
Choose a word to fill the gap in the sentence. “Gene drives are an ambitious experiment in …”. Genetics? Ecology? Evolution? Obviously, 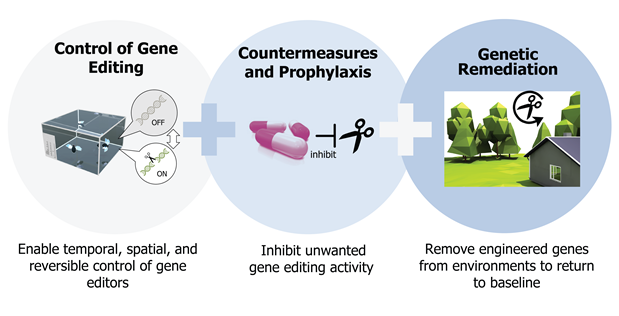
 Mosquito nets are not enough, vaccines are late to come, land reclamation in Africa is a challenge. But there is a new hope for defeating malaria, coming directly from the most advanced CRISPR frontier. The trick is a kind of genetic chain reaction fuelled by genetic elements called “gene drives”. Researchers are experimenting their power with the aim of crashing the number of mosquitoes responsible for Plasmodium transmission, by spreading genes that are bad for Anopheles gambiae. A gene behaving in Mendelian way has a 50% chance of being passed on from parent to offspring, but it can virtually reach 100% with a little help from a drive. Thus a gene designed to damage a harmful species can propagate within a few generations with a domino effect, until the population collapses. One of the founders of this futuristic strategy is an Italian molecular parasitologist:
Mosquito nets are not enough, vaccines are late to come, land reclamation in Africa is a challenge. But there is a new hope for defeating malaria, coming directly from the most advanced CRISPR frontier. The trick is a kind of genetic chain reaction fuelled by genetic elements called “gene drives”. Researchers are experimenting their power with the aim of crashing the number of mosquitoes responsible for Plasmodium transmission, by spreading genes that are bad for Anopheles gambiae. A gene behaving in Mendelian way has a 50% chance of being passed on from parent to offspring, but it can virtually reach 100% with a little help from a drive. Thus a gene designed to damage a harmful species can propagate within a few generations with a domino effect, until the population collapses. One of the founders of this futuristic strategy is an Italian molecular parasitologist: