
Don’t miss the “Draft Ethical Principles for Therapeutic Assisted Reproductive Technologies” just published by He Jiankui et al. in The CRISPR Journal. It seems the Lulu and Nana’s experiment is at odds even with their own guidelines. We are eager to hear more from He’s voice, his talk in Hong Kong is scheduled on Wednesday, 28 November 2018 (Second International Summit on Human Genome Editing-live broadcast)

 Just imagine you could find them all on the supermarket shelves, would you buy rice labeled as CRISPR or GMO, or stick to conventional non-genetically modified rice? And what price would you consider fair? Aaron Shew and colleagues from the University of Arkansas conducted a multi-country assessment of willingness-to-pay for and willingness-to-consume a hypothetical CRISPR-produced food and published their findings in Global Food Security.
Just imagine you could find them all on the supermarket shelves, would you buy rice labeled as CRISPR or GMO, or stick to conventional non-genetically modified rice? And what price would you consider fair? Aaron Shew and colleagues from the University of Arkansas conducted a multi-country assessment of willingness-to-pay for and willingness-to-consume a hypothetical CRISPR-produced food and published their findings in Global Food Security.  The European Commission’s Group of Chief Scientific Advisors has published a
The European Commission’s Group of Chief Scientific Advisors has published a 
 A proposal from the Netherlands Ministry of Infrastructure and the Environment points the way out of the GM regulatory impasse for most CRISPR crops in Europe. Rather than trying to clarify ambiguous definitions, the EU could simply amend Annex B1, that is the list of technologies that are excluded from regulation by the directive on GMOs (2001/18/EC).
A proposal from the Netherlands Ministry of Infrastructure and the Environment points the way out of the GM regulatory impasse for most CRISPR crops in Europe. Rather than trying to clarify ambiguous definitions, the EU could simply amend Annex B1, that is the list of technologies that are excluded from regulation by the directive on GMOs (2001/18/EC). 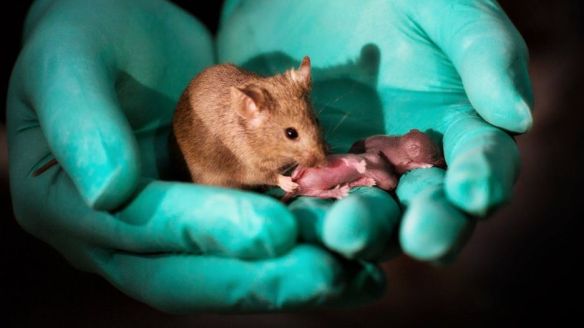
 It’s another CRISPR first: fixing a hereditary disease in utero. Scientists from Pennsylvania University and the Children’s Hospital of Philadelphia used a base editor to rescue tyrosinemia in fetal mice. “The results of this proof-of-concept work demonstrate the possibility of efficiently performing gene editing before birth, pointing to a potential new therapeutic approach for selected congenital disorders,” Avery C. Rossidis and colleagues write in
It’s another CRISPR first: fixing a hereditary disease in utero. Scientists from Pennsylvania University and the Children’s Hospital of Philadelphia used a base editor to rescue tyrosinemia in fetal mice. “The results of this proof-of-concept work demonstrate the possibility of efficiently performing gene editing before birth, pointing to a potential new therapeutic approach for selected congenital disorders,” Avery C. Rossidis and colleagues write in  “Agricultural research, or a new bioweapon system?”. This is the question asked by Guy Reeves et al. in a policy forum published in
“Agricultural research, or a new bioweapon system?”. This is the question asked by Guy Reeves et al. in a policy forum published in 