If you know the enemy and know yourself, you need not fear the result of a hundred battles. The military strategist Sun Tzu wrote it over two thousand years ago, but this quote could also apply to oncology research in the CRISPR era. Identifying the weak points of cancer cells is the first step to hit new molecular targets with the next generation of drugs.
The good news is that the Wellcome Sanger Institute has taken a giant leap toward this goal, drawing up a list of 600 candidate genes. The study just published in Nature by Mathew Garnett’s team comes with a twin paper by the Broad Institute, confirming the results by following an alternative approach. In a four-year tour de force of functional genomics, Sanger’s researchers used CRISPR to disrupt every gene in over 300 cancer models from 30 cancer types. From this amount of data, they developed a prioritization system which will guide big pharma’s hunt for new drugs.

 Do you remember the
Do you remember the 

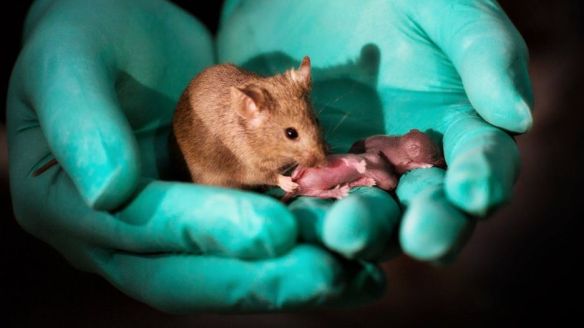
 It’s another CRISPR first: fixing a hereditary disease in utero. Scientists from Pennsylvania University and the Children’s Hospital of Philadelphia used a base editor to rescue tyrosinemia in fetal mice. “The results of this proof-of-concept work demonstrate the possibility of efficiently performing gene editing before birth, pointing to a potential new therapeutic approach for selected congenital disorders,” Avery C. Rossidis and colleagues write in
It’s another CRISPR first: fixing a hereditary disease in utero. Scientists from Pennsylvania University and the Children’s Hospital of Philadelphia used a base editor to rescue tyrosinemia in fetal mice. “The results of this proof-of-concept work demonstrate the possibility of efficiently performing gene editing before birth, pointing to a potential new therapeutic approach for selected congenital disorders,” Avery C. Rossidis and colleagues write in 


