
Many interesting papers have been published recently; here are our top three picks. They cover an innovative gene therapy trial, a new experimental approach for oncology, and the development of novel tools to map gene enhancers.
Continue reading

Many interesting papers have been published recently; here are our top three picks. They cover an innovative gene therapy trial, a new experimental approach for oncology, and the development of novel tools to map gene enhancers.
Continue reading
A scientific adventure whose ingredients include the looming threat of a fatal disease, the decision to reinvent themselves as biologists, and the goal of silencing prions.
The clinical trial with antisense oligonucleotides, born of their efforts, is considered one of the most interesting trials of 2025, but this is only a part of the story. This married couple is also pursuing other avenues to halt the onset of prion diseases. In the summer of 2024, they published a study in Science using epigenetic editing in mice. Then, in January 2025, their experiments with base editing were published in Nature Medicine. Yet Sonia Vallabh was a newly graduated jurist, and her husband, Eric Minikel, was working in urban planning, when they discovered that she carried a mutation that would condemn her to die of fatal familial insomnia within two or three decades.
Continue reading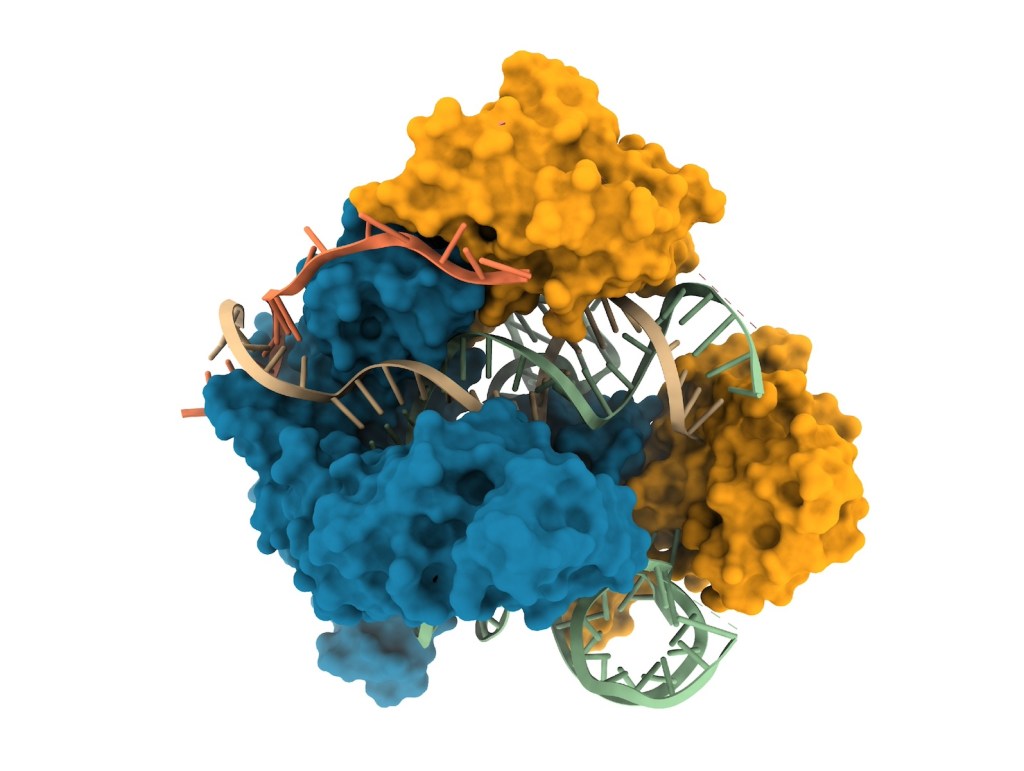
It is currently only a preprint on bioRxiv, but it has already attracted significant attention from the scientific community and the journal Science. Mammoth Biosciences, a company founded by CRISPR co-inventor Jennifer Doudna, has developed NanoCas, a mini-editor that is just one-third the size of traditional gene-editing scissors (Cas9).
Continue reading
Have you read Jennifer Doudna’s article in Wired? She discusses how the combination of CRISPR and artificial intelligence could be transformational. In her lab, researchers recently used AI tools to help find “small gene-editing proteins that had been sitting undiscovered in public genome databases because we simply didn’t have the ability to crunch all of the data that we’ve collected”.
Continue reading
Experimental patients often find themselves in a paradoxical situation: they must be sick enough to qualify for a clinical trial but healthy enough to endure its side effects. They also need the audacity to subject their bodies to protocols whose safety and efficacy remain unproven. For this reason, many describe them as pioneers or even warriors.
Continue reading
It’s been about a year since the first CRISPR-based treatment was approved in the United States and Europe. However, those expecting a surge in approvals of new gene-editing therapies may be disappointed. Next in line will likely be another approach to treating sickle cell disease, followed by therapies for TTR amyloidosis and hereditary angioedema around 2026-27. According to The CRISPR Journal, that’s all we can expect over the next 3-5 years. Is Casgevy destined to stand out like a cathedral in the desert? We have a super-versatile platform capable of fixing a myriad of genetic defects, so why is the CRISPR revolution slowing down? To understand the looming crisis and the countermeasures needed, don’t miss Fyodor Urnov’s in-depth editorial entitled “Give Cas a Chance: An Actionable Path to a Platform for CRISPR Cures.”

This photo shows the first American “non-experimental” patient leaving the hospital after completing the CRISPR-based treatment for sickle cell anemia (Casgevy). The New York Times detailed this “official first,” which followed the success of a clinical trial involving dozens of patients like Victoria Gray. We still know little about the first person who is beginning treatment in Europe since this therapy became an “approved drug”. According to Osservatorio Terapie Avanzate he is a young adult (23 years), who arrived in Italy in 2014 and living in the Umbria region, where is being also treated. Undergoing cell extraction and reinfusion of edited cells is an invasive and exhausting process, but now the American Kendric Cromer (12 years old) and other “first patients” can hope to lead full lives—without painful crises or blood transfusions. Best of luck!

MicroRNAs won Victor Ambros and Gary Ruvkun the 2024 Nobel Prize in Medicine. Thomas Cech (Nobel Laureate for the discovery of catalytic RNA) has found a fun way to explain how they work. His book, which I reviewed a few weeks ago, is a mine of insights and information. Here is a small excerpt.
Continue readingSammy Basso, 28, the longest-living person with progeria and a brilliant mind, has passed away. He was a biologist passionate about genome editing, and we’d like to remember him through this dialogue with David Liu.
Uditi Saraf died before receiving treatment, but efforts launched for her could help spell a happy ending for other patients awaiting advanced life-saving therapies

Familial encephalopathy with neuroserpin inclusion bodies is a rare neurodegenerative disease with no cure due to the accumulation of toxic proteins in the brain. Depending on the specific mutation, the age of onset can vary greatly. In Uditi Saraf’s case, the first symptoms started early, at age 9. As she worsened, her parents decided to have her genome sequenced, identifying the genetic defect and diagnosing the condition. Their race against time to try to save their daughter was chronicled in an article in Nature, which also offers a glimpse into India’s efforts to make genomic treatments more accessible (see also Nature Biotechnology on gene and cell therapies in the Global South).