Food for Thought is a coalition of 18 Italian associations in the agrifood sector, established in 2017 to promote innovation in agriculture. Today, nearly eight years after the first manifesto, a new one has been presented to address the challenges of both the present and the future, including the climate crisis, geopolitical tensions, and changing consumption patterns.
The event was held yesterday in Rome at the initiative of Senator Bartolomeo Amidei, as part of the activities of the Parliamentary Intergroup on Made in Italy and Innovation.
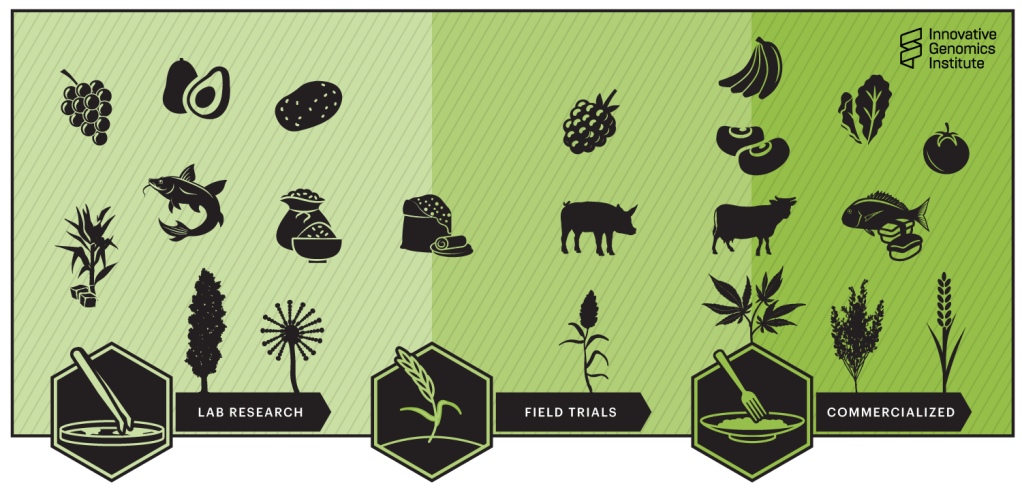
Among the key points is the inclusion of New Genomic Techniques — a clear sign that there is widespread awareness within the Italian productive sector of the importance of genetic improvement, and that there are voices in the national Parliament willing to advocate for “innovative and sustainable agriculture.”