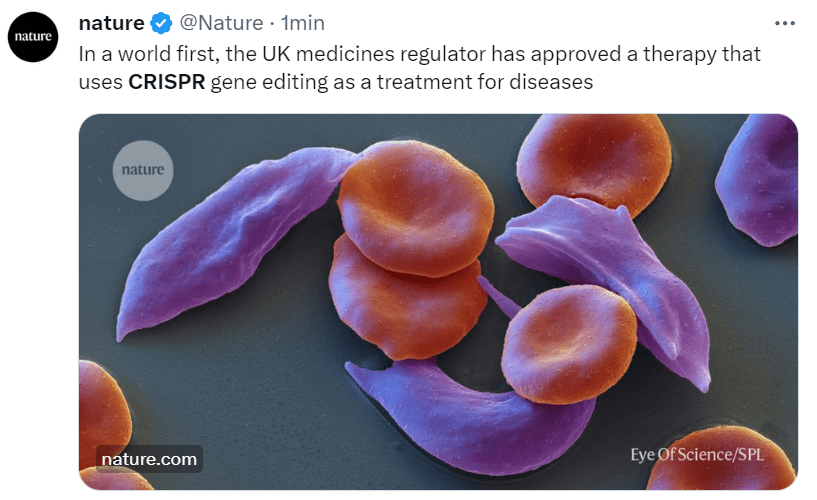
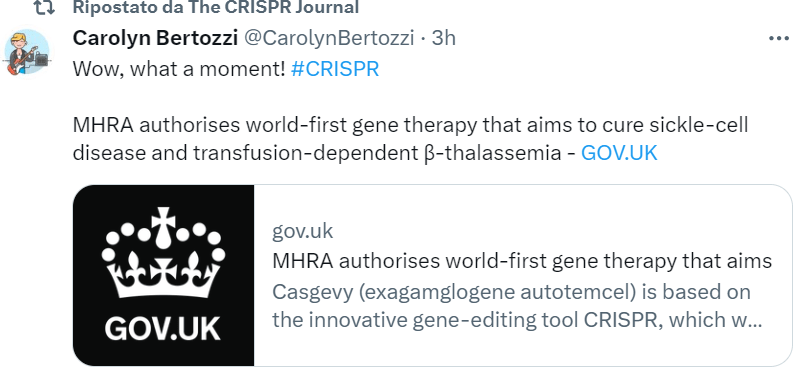
The New Genomic Techniques (NGTs) such as CRISPR are slowly rearranging the ranks in the debate on plant genetics and GM food. A few stakeholders that have always been hostile to GMOs have decided to open the door to the new opportunities coming from gene editing (in Italy, for example, the powerful farmers’ association Coldiretti has changed its mind). Others (e.g., Greenpeace, despite some isolated and courageous vanguards) have confirmed an ideological/cultural opposition, even in cases where the genetic intervention is so soft that the plants are concretely indistinguishable from those developed by conventional techniques. As for the galaxy of organic farming, it is standing still, yet something is moving. Don’t miss the article published in EUobserver by Lone Andersen and other organic farmers, who are calling on European institutions not to ban the new breeding techniques from their fields in the ongoing regulatory revision. “By encouraging the use of a limited share of the new NGTs in organic agriculture while preserving the non-GM status, the EU can further promote sustainable practices and reinforce its commitment to a greener future,” they write.